VSEPR Theory
|
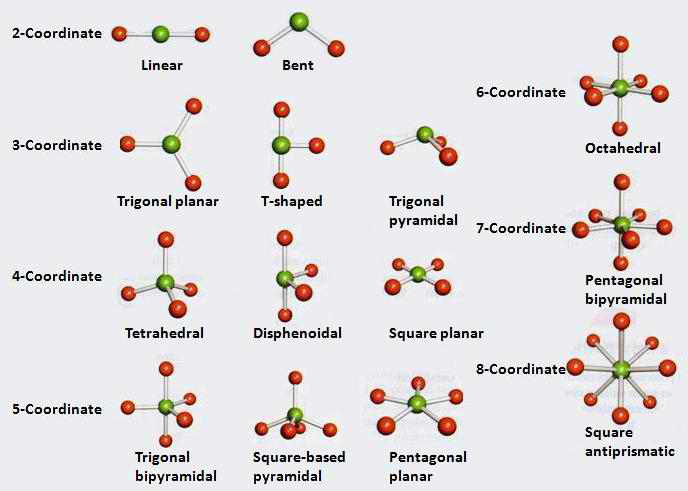
Dr. Gillespie's greatest contribution to modern science lies in his development of the VSEPR theory with his partner Ronald Nyholm. His theory states that the valence electrons of an atom are positioned in such a way that their potential energies due to their respective distances are at a minimum; that is, they are as far away from each other as possible. Due to this discovery, Dr. Gillespie was able to determine the location of electron bonds in molecules, that is, their appearances. This includes the theoretical prediction of bond angles between any three atoms in a molecule. The experimental results from the bond angles led to the quantum mechanical explanation of hybridization in bonds, a major step forward in this relatively new field of science.
|
Determining the Covalent Radius of Fluorine
Dr. Gillespie has proved to the scientific community his theoretical prowess through determining the covalent radius of fluorine. The values of covalent radii of atoms are usually determined by experimental results, but finding the covalent radius of fluorine proved to be a difficult task due to its high electronegativity and small atomic radius. Through interpretations of different values of the covalent radii of fluorine when bonded with other atoms, Dr. Gillespie theoretically predicted the covalent radius of fluorine, when experiments couldn't even find a result.